The Math Science. This chemistry video tutorial focuses on the root mean square velocity equation.

Kinetic Molecular Theory Of Gases And Root Mean Square Speed Calculating Gas Ke Speed Root Mean Square Molecular Kinetic Energy
Root mean square velocity RMS valueis the square root of the mean of squares of the velocity of individual gas molecules.

Formula for root mean square speed. The formula for the root-mean-squared speed is as follows. Online chemistry calculator to calculate root mean square RMS speed of gas using gas molecular mass value. Here we will learn a simple method to derive the Root Mean Square speed.
RMSE sqrtfracsum_i1nX_obsi-X_modeli2n Where x obs is observed values x model is modelled values at time i. U 3 R T M U 3 R T M. Root mean square speed 3RTM The most probable velocity The most probable velocity at which the most molecules in gas travel.
The Root-Mean-Square Speed formula for the molecular speed of gaseous particles is. This speed is then referred to as the root-mean-square speed v rms. R gas constant 83145 Jmol 1 K 1.
Since number of molecules is very high hence we use Maxwells distribution method. T Temperature in Kelvin. M Molar mass of the gas Kgmole R Molar gas constant.
In order to obtain the dimension of a speed the square root of this mean value must be calculated. Compute the Temperature of gas based on the Root-Mean-Square formula. However this can be automatically converted to other velocity units via the pull-down menu.
The root-mean-square speed of molecules is the speed at which all the molecules have the same total kinetic energy as in case of their actual speed. The variable of integration velocity can attain all possible values. The calculator returns the velocity V rms in meters per second.
Gas Mean Velocity V rms. The average velocity of gas particles is found using the root mean square velocity formula. It is represented by the equation.
Root Mean Square Error Formula. It gives an example practice. RMS speed of H 2 He N 2 O 2 and CO 2 gases at 310 K temperature.
Latexv_rmssqrtfrac3RTMlatex where v rms is the root-mean-square of the velocity M m is the molar mass of the gas in kilograms per mole R is the molar gas constant and T is the. It gives an example practice problem showing you how to calculate the roo. Root mean square speed Vrms is defined as the square root of the mean of the square of speeds of all molecules.
U is the molecular speed of gas particles. Derivation of root mean square speed. The root-mean-square speed is the square root of the mean of the squares of the velocities.
V r m s 3 R T M V r m s 3 R T M. The RMSE of a predicted model with respect to the estimated variable x model is defined as the square root of the mean squared error. Compute the molecular speed of a gas based on the Root-Mean-Square formula.
The root-mean-square speed u is the square root of the average speeds of the molecules in a sample of gas at a specific temperature and pressure. This quantity is defined so that addition of squares of velocities will not give zero unlike average. rms 3RTM rms root mean square velocity in msec.
It is defined as the square root of the average velocity-squared of the molecules in a gas. Root-mean-square speed is the measure of the speed of particles in a gas that is most convenient for problem solving within the kinetic theory of gases. R-Gas Constant with SI Units.
The root mean square velocity RMS velocity is a way to find a single velocity value for the particles. It is denoted by vrms v2. Root mean square speed vrms is defined as the square root of the mean of the square of speeds of all molecules.
The average speed of molecules can be calculated as an integral of the Maxwell-Boltzmann distribution function multiplied by the magnitude of velocity of a molecule v. The Math Science. Vrms v2.
V rms Root-mean-square velocity. The first distinctive velocity is easiest to estimate and term as the most probable velocity. The root-mean-square speed is the measure of the speed of particles in a gas defined as the square root of the average velocity-squared of the molecules in a gas.

Secret Of Fast Math Revealed Learn How To Calculate Faster Than The Speeding Calculator Math Tricks Math Methods Simple Math

Ideal Gas Law Problems And Solutions Boyle S Low Charl S Low Combination Of Boyle S Low And Charl S Low I Ideal Gas Law Problem And Solution Kinetic Theory

Standard Deviation Google Search Standard Deviation Root Mean Square Arithmetic

Assumptions And Formulation Of Kinetic Theory Of Gas Derivation Of Kinetic Gas Equation Root Mean Square Velo Study Chemistry Root Mean Square

Rms Value Average Value Peak Value Peak Factor Form Factor In Ac Root Mean Square Ohms Law Rms

591 Me Gusta 2 Comentarios Chavan S Education Hub Thechavanclasses En Instagram Root Mean Square Velocity Do Follow Thechavanclasses Chem

Average Speed Is Determined By Applying The Formula Total Distance Travelled Total Time Taken Average Speed Will Arithmetic Formula How To Apply Teaching

Rms Value Average Value Peak Value Peak Factor Form Factor In Ac Engineering Notes Root Mean Square Rms

Free Study Materials For Competitive Exams Tricks To Solve Train Problems Math Problem Solving Math Tricks Solving

Rms Value Average Value Peak Value Peak Factor Form Factor In Ac Root Mean Square Ohms Law Rms

Rms Value Average Value Peak Value Peak Factor Form Factor In Ac Ohms Law Root Mean Square Sine Wave

Rms Value Average Value Peak Value Peak Factor Form Factor In Ac Root Mean Square Ohms Law Free Math Resources
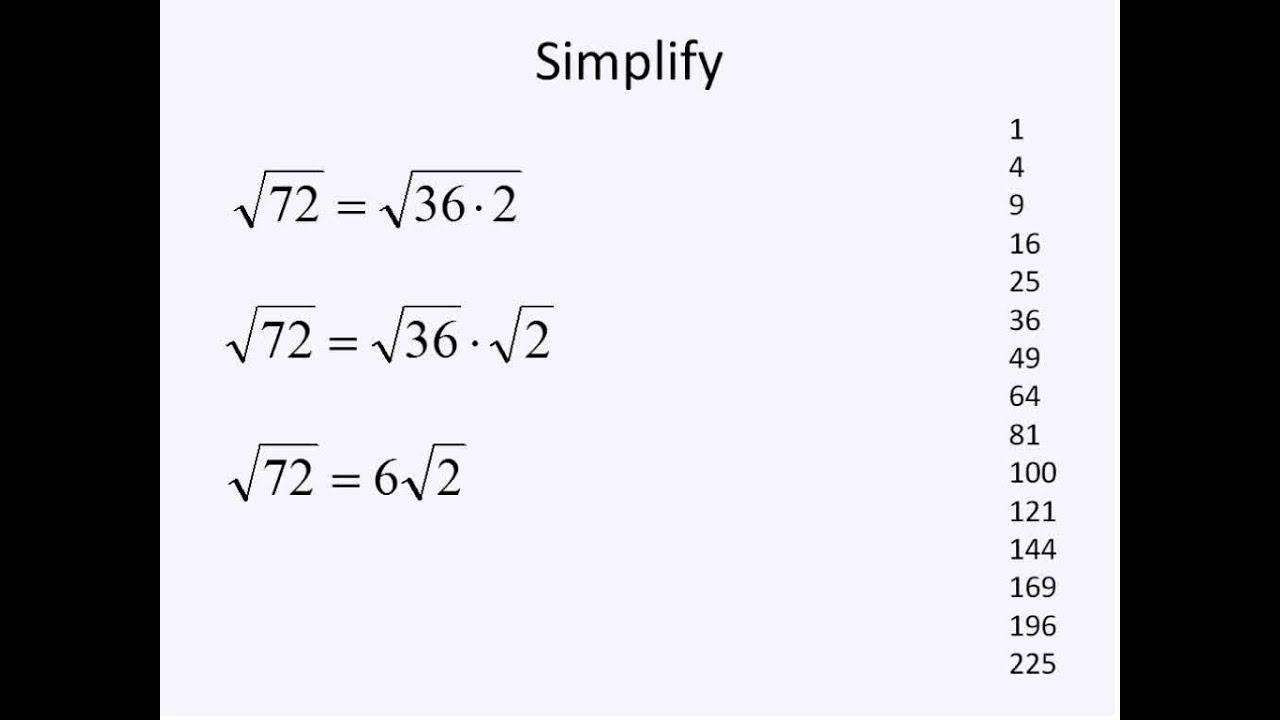
Irrational Square Roots Simplifying Math Evaluating Algebraic Expressions Literal Equations Radical Expressions

Kinetic Gas Equation Kinetic Theory Study Chemistry Root Mean Square

Mit Numerical Methods For Pde Lecture 3 Finite Difference For 2d Poisson S Equation Numerical Methods Lecture Method

Radius Of Gyration In 2021 Radii Root Mean Square General Physics




